The oncology industry is undergoing a transformative shift driven by the limitations of existing therapies, including immune checkpoint inhibitors like Keytruda (pembrolizumab). While such drugs have demonstrated groundbreaking results in some patients, a substantial proportion—up to 70~85% in certain indications—either do not respond or relapse due to intrinsic or acquired resistance.
Immunotherapy Resistance: A Growing Bottleneck
The FDA and EMA are prioritizing therapies that address these non-responsive populations through breakthrough and fast-track designations.
Palade has developed “MoSTT-CelSuR” (Molecular Screening Technology Targeting a Cell Surface Receptor) technology, a highly accurate and efficient high-throughput screening platform technology for molecules that activate (agonists) or inhibit (antagonists) a GPCR (G protein-coupled receptor).
GPCRs are a major target of drugs for diverse human diseases including diabetes, obesity, asthma, cardiovascular diseases, pain, CNS diseases and cancer. 30~40% of FDA-approved drugs target GPCRs. As of 2025, there are approximately 60~70 GPCR-target blockbuster drugs that include Ozempic, Wegovy, Singulair, and morphine.
Related with Keytruda and cancer immunotherapy, recent findings identified that GPCRs are major players to control tumor microenvironment and to determine the responsiveness of Keytruda.
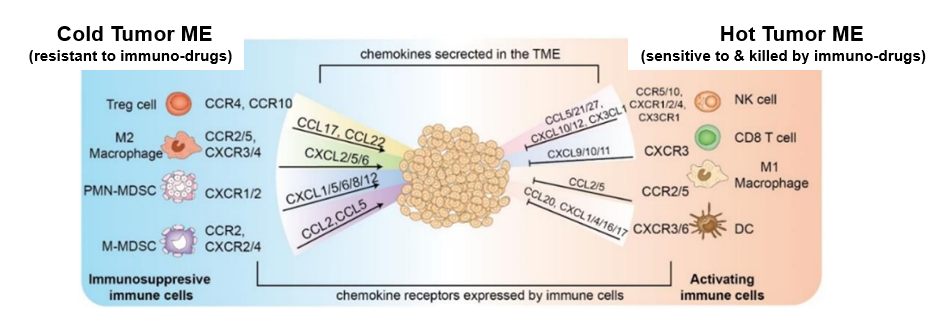
Keytruda responsiveness is determined by the tumor microenvironment (TME) of tumor-protective cold TME versus tumor-attacking hot TME.
GPCRs play key roles in controlling cold TME (composed of tumor-protective Treg, MDSC, CAF and TAM2 cells) and hot TME (composed of tumor-attaching CD4 or CD8 T cells, NK, DC, M1 cells). Chemokines and their associated GPCRs control the nature of immune cells infiltrating the tumor microenvironment (TME). Well-established findings are that chemokines, CXCL9, CXCL10 and CXCL11, bind the CXCR3 GPCR on T cells to promote T cell migration to the tumor. T cell dysfunction and exhaustion are also closely related with GPCRs.
Tumor microenvironment-specific conditions such as hypoxia and acidity activate a specific set of GPCRs on immune cells to inhibit the function of the immune cells, leading to immuno-suppressive conditions.
Palade BioTech develop GPCR-target tumor microenvironment modulator drugs that convert the cold TME to hot TME, which promotes the function of Keytruda and cancer immunotherapy.
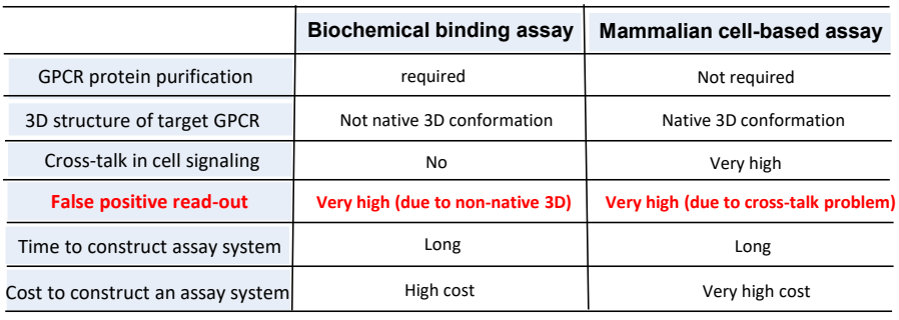
Development of GPCR-target drugs are extremely difficult. Two main drug screening methods (biochemical binding assays & mammalian cell-based functional assays) have the problem of high false-positive readouts that results in less than 10% success rates of drug development from the initial hit compounds.
The 7 transmembrane structure of GPCR makes biochemical binding assays difficult to contain the native form of GPCR. The frequent cross-talks among GPCRs (approximately 900 GPCRs in humans) makes mammalian cell-based functional assays difficult to read out the correct signal.
Two main drug screening methods have the problem of high false-positive readouts
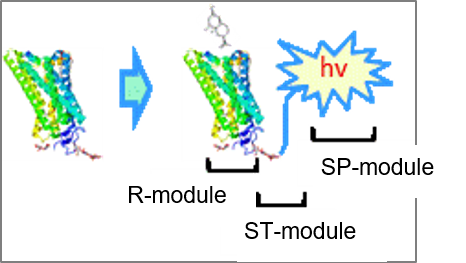
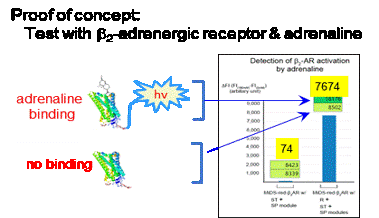
With a bold vision to become a global leader in resistance-targeted therapeutics, we are pioneering curative solutions for drug-resistant, immunotherapy-refractory cancers and incurable viral threats, the areas of highest mortality and unmet need.