Cytokines (interferons and interleukins) regulate the function of T cells and immune cells like Keytruda. Cytokines were identified long before PD-1 and PD-L1, immune checkpoint regulators that Keytruda targets. As certain cytokines such as IL-2, IL-12 and IL-15 can activate T cells and immune cells, these cytokines can facilitate to convert cold tumor microenvironment (TME) to hot TME. Therefore, in principle, these cytokines can be developed as cancer immunotherapy drugs.
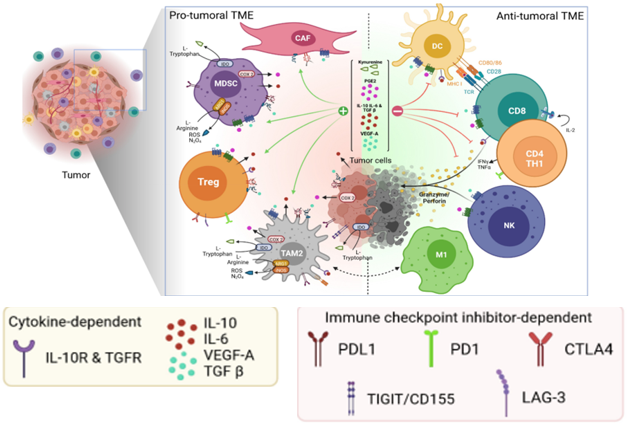
(Modified from https://www.tandfonline.com/doi/epdf/10.1080/2162402X.2022.2120676?needAccess=true)
However, cytokines were turned out to be very difficult to be developed as cancer drugs because a systemic administration of cytokine is very toxic as it engenders systemic toxic immune reactions such as cytokine storms.
High doses of cytokine required for cancer treatment distributes broadly and binds to receptors in many non-target tissues, whereby activating non-target immune cells and engendering cytokine storm and serious toxicity.
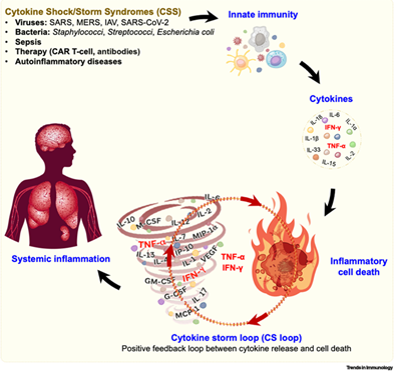
(Adopted from https://www.cell.com/action/showPdf?pii=S1471-4906%2821%2900115-0)
As tumor-specific targeting antibody cancer drugs such as Keytruda have been immensely successful in cancer treatment, drug developers started recently to develop immunocytokine cancer drugs that combine antibody and cytokine. Cytokine can be targeted by antibody to tumor-specific tissues without activating immune cells in non-target tissues. This can reduce the toxicity/cytokine storm problem of cytokines when they are administered as single molecule.
Furthermore, by combining anti-cancer antibody like Keytruda with cytokine that can convert cold tumor microenvironment (TME) to hot TME, immunocytokine cancer drugs can have stronger anti-cancer activity than anti-cancer antibody alone.
Recently, there are several encouraging pre-clinical and early clinical results reported by immunocytokine developers.
This situation prompts to expect “the next-generation Keytruda” to come out in the market soon.
Key to the development of the next-generation immunocytokine” is how to control the activity of cytokine in antibody-cytokine fusion protein. Cytokine has to remain as inactive (receptor non-binding) until its fusion partner antibody binds to the targeted tumor tissue. After reach to the targeted tumor tissue, the cytokine now has to bind and activate its receptor on the tumor tissue.
Palade Bio develop P-AID (Palade Advanced Immunocytokine Drug) using STarMIACAT (Surface Targeting Mighty Inactive Agonist and Controlled Activation Technology), an innovative cytokine activity-controlling technology. The STarMIACAT technology can target cytokines in a concentrated way to tumor tissues and can activate upon they reach to the target tissue.
As STarMIACAT can deliver and activate more concentrated cytokine to tumor tissues than conventional technologies that other companies adopt, P-AID cancer drugs will have better chances to be developed as “the next-generation Keytruda”.
With a bold vision to become a global leader in resistance-targeted therapeutics, we are pioneering curative solutions for drug-resistant, immunotherapy-refractory cancers and incurable viral threats, the areas of highest mortality and unmet need.